______________
The impact of Code𝛘 on the physicochemical properties and 3D organization of the peritrophic matrix from the model pest beetle Tribolium castaneum
About the Project
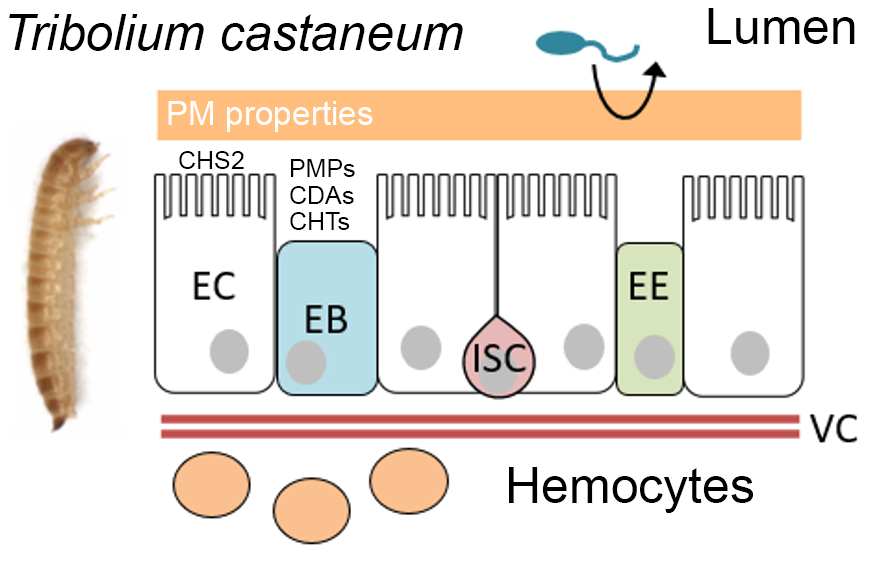
The midgut of most insects is lined by a semipermeable, extracellular lining called peritrophic matrix (PM), which has several important functions such as improving the intestinal performance, generating a barrier against intestinal pathogens and parasites, and protecting from mechanical and chemical damage. More recently, research has also focused on the PM’s role in gut innate immunity and insect-pathogen interactions. Understanding all these different functions requires a deeper insight into PM composition and into the construction principles of its 3D organization. The PM is composed of varying constituents that confer characteristic properties needed for the respective function. Next to chitin, a polymer of N-acetylglucosamine (GlcNAc), which appears to be deacetylated to some extent, various chitin-associated PM proteins (PMPs), some of them extensively glycosylated, are considered major structural elements of the PM. It still remains speculative how the different components of the PM exactly interact and in which way they influence each other. In addition, the roles of chitin modification by deacetylases and chitinases in organizing the 3D structure of the PM are largely unknown. We hypothesize that the degree of deacetylation and polymerization as well as patterns of deacetylation (which we term Codec), written by chitin deacetylases and chitinases and recognized by PMPs, have a major impact on physicochemical properties and 3D organization of the PM in different midgut regions and thus affect the barrier function. In this project, we will analyze different PM constituents in the coleopteran pest model Tribolium castaneum. We will focus on the structure-function relationships of chitin fibrils and PM proteins (PMPs), which have different types and numbers of chitin binding domains (CBDs). We aim to understand how PMPs help to organize the PM’s fibrillary network, so that a permeability barrier with decreasing exclusion sizes from anterior to posterior midgut regions is generated, as we have demonstrated it in larvae of T. castaneum. We will determine which type of chitin isomorph is present in the PM at different midgut regions, decipher the function of midgut-specific chitin deacetylases and chitinases, and examine the binding mode of different PMPs and their function in 3D organizing chitin fibrils. Finally, we will elucidate probable differences in binding affinities and selectivities between the various types of CBDs that are found in PMPs in different numbers. In doing so, we aim to explain how PMPs organize the 3D network in the PM and control its different properties along the midgut. This will finally provide fundamental knowledge for the development of active compounds targeting the PM for pest control.

Principal Investigators

Prof. Dr. Hans Merzendorfer
University of Siegen
Department Chemistry-Biology
Molecular Biology
57068 Siegen
